

實驗室前期對電化學沉積Cu2O薄膜進行了系統(tǒng)的研究,發(fā)現(xiàn)其沉積與沉積電壓火诸、溶液溫度和pH值等密切相關锦针。本文以電化學沉積Cu2O薄膜為例,從而在實驗室構建橢偏儀在位監(jiān)控電化學沉積系統(tǒng)置蜀。
展示全部 
橢偏儀在位表征電化學沉積的系統(tǒng)搭建(四)-電化學沉積及原理
2.電化學沉積
電化學沉積是半導體薄膜沉積和微電子制備銅互連的重要制備方法奈搜。而在沉積過程中的成核和生長對于半導體薄膜和銅互連的性質非常重要,橢偏儀在位監(jiān)測提供一種實時監(jiān)控薄膜沉積的方法盯荤。但是橢偏儀在位監(jiān)測受到光路設計馋吗,實驗裝置,固液界面以及光譜解析的影響秋秤,構建其監(jiān)測系統(tǒng)是一個挑戰(zhàn)宏粤。實驗室前期對電化學沉積Cu2O薄膜進行了系統(tǒng)的研究,發(fā)現(xiàn)其沉積與沉積電壓灼卢、溶液溫度和pH值等密切相關绍哎。本文以電化學沉積Cu2O薄膜為例,從而在實驗室構建橢偏儀在位監(jiān)控電化學沉積系統(tǒng)鞋真。
不同于真空薄膜生長崇堰,電化學沉積生長過程涉及到溶液層和固液界面,導致其在位監(jiān)測是一個挑戰(zhàn)涩咖。
2.1原理
電化學沉積是利用氧化還原反應在電極表面上沉積得到各種薄膜的材料制備方法赶袄。在沉積過程中電極表面的狀態(tài)、沉積電壓或電流的大小抠藕、沉積電解液的溫度和pH值都會對得到的薄膜的相產(chǎn)生影響饿肺。故而可以通過沉積中電壓、電流的調控沉積不同成分組成及不同微觀形貌的薄膜盾似。利用電化學沉積可以減小制作成本敬辣、提高產(chǎn)量,且由于其較好的可控性和可操作性零院,目前已經(jīng)廣泛應用于工業(yè)化生產(chǎn)溉跃,實現(xiàn)電化學大規(guī)模沉積。
電化學薄膜沉積可分為恒壓沉積和恒電流沉積告抄。恒壓法又分為過電位沉積和欠電位沉積撰茎。過電位沉積就是在大于能斯特電位的電壓下進行沉積。欠電位沉積除了在單層(亞單層)沉積外打洼,有時當基底影響到第二龄糊、第三個單層時也可以在欠電位下沉積逆粹。Hevesy于1912年第1次報道了不同放射性元素在Cu電極上的欠電位沉積現(xiàn)象。目前炫惩,報道過的欠電位沉積體系有質子性溶劑僻弹、離子液體及有機溶劑等。
在電化學過程中他嚷,由于電化學反應的存在電極和溶液界面會出現(xiàn)溶劑離子濃度從本體溶液濃到電極界面濃度降低的過程蹋绽,而從本體溶液濃度到電極表面溶液濃度的這一過渡區(qū)域就叫擴散層。圖1-6為Gouy-Chapman-Stern雙電層模型筋蓖,擴散層是外亥姆霍茲層(OHP)到溶劑濃度達本體溶液濃度的區(qū)域卸耘,擴散層的厚度取決于溶液中離子的濃度,當濃度大于10-2M時粘咖,擴散層的厚度將小于30nm鹊奖。
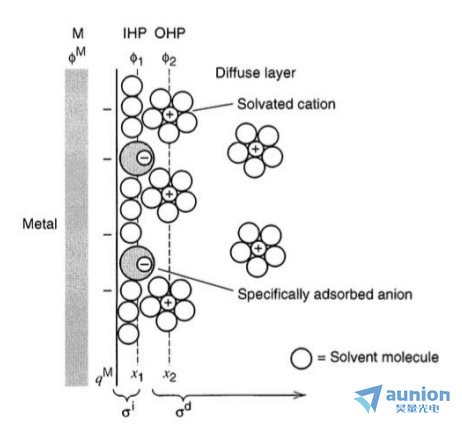
圖1-6電極-溶液雙層區(qū)模型
因此電化學沉積過程發(fā)生在固液界面,而溶液的固液界面比較復雜涂炎,包含了擴散層等忠聚。
了解更多橢偏儀詳情,請訪問上海昊量光電的官方網(wǎng)頁:
http://www.wjjzl.com/three-level-56.html
更多詳情請聯(lián)系昊量光電/歡迎直接聯(lián)系昊量光電
關于昊量光電:
上海昊量光電設備有限公司是光電產(chǎn)品專業(yè)代理商唱捣,產(chǎn)品包括各類激光器两蟀、光電調制器、光學測量設備震缭、光學元件等赂毯,涉及應用涵蓋了材料加工、光通訊拣宰、生物醫(yī)療党涕、科學研究、國防巡社、量子光學膛堤、生物顯微、物聯(lián)傳感晌该、激光制造等肥荔;可為客戶提供完整的設備安裝,培訓朝群,硬件開發(fā)燕耿,軟件開發(fā),系統(tǒng)集成等服務姜胖。
您可以通過我們昊量光電的官方網(wǎng)站www.wjjzl.com了解更多的產(chǎn)品信息誉帅,或直接來電咨詢4006-888-532。
相關文獻:
[1] WONG H S P, FRANK D J, SOLOMON P M et al. Nanoscale cmos[J]. Proceedings of the IEEE, 1999, 87(4): 537-570.
[2] LOSURDO M, HINGERL K. ellipsometry at the nanoscale[M]. Springer Heidelberg New York Dordrecht London. 2013.
[3] DYRE J C. Universal low-temperature ac conductivity of macroscopically disordered nonmetals[J]. Physical Review B, 1993, 48(17): 12511-12526. DOI:10.1103/PhysRevB.48.12511.
[4] CHEN S, KüHNE P, STANISHEV V et al. On the anomalous optical conductivity dISPersion of electrically conducting polymers: Ultra-wide spectral range ellipsometry combined with a Drude-Lorentz model[J]. Journal of Materials Chemistry C, 2019, 7(15): 4350-4362.
[5] 陳籃,周巖. 膜厚度測量的橢偏儀法原理分析[J]. 大學物理實驗, 1999, 12(3): 10-13.
[6] ZAPIEN J A, COLLINS R W, MESSIER R. Multichannel ellipsometer for real time spectroscopy of thin film deposition from 1.5 to 6.5 eV[J]. Review of Scientific Instruments, 2000, 71(9): 3451-3460.
[7] DULTSEV F N, KOLOSOVSKY E A. Application of ellipsometry to control the plasmachemical synthesis of thin TiONx layers[J]. Advances in Condensed Matter Physics, 2015, 2015: 1-8.
[8] DULTSEV F N, KOLOSOVSKY E A. Application of ellipsometry to control the plasmachemical synthesis of thin TiONx layers[J]. Advances in Condensed Matter Physics, 2015, 2015: 1-8.
[9] YUAN M, YUAN L, HU Z et al. In Situ Spectroscopic Ellipsometry for Thermochromic CsPbI3 Phase Evolution Portfolio[J]. Journal of Physical Chemistry C, 2020, 124(14): 8008-8014.
[10] 焦楊景.橢偏儀在位表征電化學沉積的系統(tǒng)搭建.云南大學說是論文,2022.
[11] CANEPA M, MAIDECCHI G, TOCCAFONDI C et al. Spectroscopic ellipsometry of self assembLED monolayers: Interface effects. the case of phenyl selenide SAMs on gold[J]. Physical Chemistry Chemical Physics, 2013, 15(27): 11559-11565. DOI:10.1039/c3cp51304a.
[12] FUJIWARA H, KONDO M, MATSUDA A. Interface-layer formation in microcrystalline Si:H growth on ZnO substrates studied by real-time spectroscopic ellipsometry and infrared spectroscopy[J]. Journal of Applied Physics, 2003, 93(5): 2400-2409.
[13] FUJIWARA H, TOYOSHIMA Y, KONDO M et al. Interface-layer formation mechanism in (formula presented) thin-film growth studied by real-time spectroscopic ellipsometry and infrared spectroscopy[J]. Physical Review B - Condensed Matter and Materials Physics, 1999, 60(19): 13598-13604.
[14] LEE W K, KO J S. Kinetic model for the simulation of hen egg white lysozyme adsorption at solid/water interface[J]. Korean Journal of Chemical Engineering, 2003, 20(3): 549-553.
[15] STAMATAKI K, PAPADAKIS V, EVEREST M A et al. Monitoring adsorption and sedimentation using evanescent-wave cavity ringdown ellipsometry[J]. Applied Optics, 2013, 52(5): 1086-1093.
[16] VIEGAS D, FERNANDES E, QUEIRóS R et al. Adapting Bobbert-Vlieger model to spectroscopic ellipsometry of gold nanoparticles with bio-organic shells[J]. Biomedical Optics Express, 2017, 8(8): 3538.
[17] ARWIN H. Application of ellipsometry techniques to biological materials[J]. Thin Solid Films, 2011, 519(9): 2589-2592.
[18] ZIMMER A, VEYS-RENAUX D, BROCH L et al. In situ spectroelectrochemical ellipsometry using super continuum white laser: Study of the anodization of magnesium alloy [J]. Journal of Vacuum Science & Technology B, 2019, 37(6): 062911.
[19] ZANGOOIE S, BJORKLUND R, ARWIN H. Water Interaction with Thermally Oxidized Porous Silicon Layers[J]. Journal of The Electrochemical Society, 1997, 144(11): 4027-4035.
[20] KYUNG Y B, LEE S, OH H et al. Determination of the optical functions of various liquids by rotating compensator multichannel spectroscopic ellipsometry[J]. Bulletin of the Korean Chemical Society, 2005, 26(6): 947-951.
[21] OGIEGLO W, VAN DER WERF H, TEMPELMAN K et al. Erratum to ― n-Hexane induced swelling of thin PDMS films under non-equilibrium nanofiltration permeation conditions, resolved by spectroscopic ellipsometry‖ [J. Membr. Sci. 431 (2013), 233-243][J]. Journal of Membrane Science, 2013, 437: 312..
[22] BROCH L, JOHANN L, STEIN N et al. Real time in situ ellipsometric and gravimetric monitoring for electrochemistry experiments[J]. Review of Scientific Instruments, 2007, 78(6).
[23] BISIO F, PRATO M, BARBORINI E et al. Interaction of alkanethiols with nanoporous cluster-assembled Au films[J]. Langmuir, 2011, 27(13): 8371-8376.
[24] 李廣立. 氧化亞銅薄膜的制備及其光電性能研究[D]. 西南交通大學, 2016.
[25] 董金礦. 氧化亞銅薄膜的制備及其光催化性能的研究[D]. 安徽建筑大學, 2014.
[26] 張楨. 氧化亞銅薄膜的電化學制備及其光催化和光電性能的研究[D]. 上海交通大學材料科 學與工程學院, 2013.
[27] DISSERTATION M. Cellulose Derivative and Lanthanide Complex Thin Film Cellulose Derivative and Lanthanide Complex Thin Film[J]. 2017.
[28] NIE J, YU X, HU D et al. Preparation and Properties of Cu2O/TiO2 heterojunction Nanocomposite for Rhodamine B Degradation under visible light[J]. ChemistrySelect, 2020, 5(27): 8118-8128.
[29] STRASSER P, GLIECH M, KUEHL S et al. Electrochemical processes on solid shaped nanoparticles with defined facets[J]. Chemical Society Reviews, 2018, 47(3): 715-735.
[30] XU Z, CHEN Y, ZHANG Z et al. Progress of research on underpotential deposition——I. Theory of underpotential deposition[J]. Wuli Huaxue Xuebao/ Acta Physico - Chimica Sinica, 2015, 31(7): 1219-1230.
[31] PANGAROV n. Thermodynamics of electrochemical phase formation and underpotential metal deposition[J]. Electrochimica Acta, 1983, 28(6): 763-775.
[32] KAYASTH S. ELECTRODEPOSITION STUDIES OF RARE EARTHS[J]. Methods in Geochemistry and Geophysics, 1972, 6(C): 5-13.
[33] KONDO T, TAKAKUSAGI S, UOSAKI K. Stability of underpotentially deposited Ag layers on a Au(1 1 1) surface studied by surface X-ray scattering[J]. Electrochemistry Communications, 2009, 11(4): 804-807.
[34] GASPAROTTO L H S, BORISENKO N, BOCCHI N et al. In situ STM investigation of the lithium underpotential deposition on Au(111) in the air- and water-stable ionic liquid 1-butyl-1-methylpyrrolidinium bis(trifluoromethylsulfonyl)amide[J]. Physical Chemistry Chemical Physics, 2009, 11(47): 11140-11145.
[35] SARABIA F J, CLIMENT V, FELIU J M. Underpotential deposition of Nickel on platinum single crystal electrodes[J]. Journal of Electroanalytical Chemistry, 2018, 819(V): 391-400.
[36] BARD A J, FAULKNER L R, SWAIN E et al. Fundamentals and Applications[M]. John Wiley & Sons, Inc, 2001.
[37] SCHWEINER F, MAIN J, FELDMAIER M et al. Impact of the valence band structure of Cu2O on excitonic spectra[J]. Physical Review B, 2016, 93(19): 1-16.
[38] XIONG L, HUANG S, YANG X et al. P-Type and n-type Cu2O semiconductor thin films: Controllable preparation by simple solvothermal method and photoelectrochemical properties[J]. Electrochimica Acta, 2011, 56(6): 2735-2739.
[39] KAZIMIERCZUK T, FR?HLICH D, SCHEEL S et al. Giant Rydberg excitons in the copper oxide Cu2O[J]. Nature, 2014, 514(7522): 343-347.
[40] RAEBIGER H, LANY S, ZUNGER A. Origins of the p-type nature and cation deficiency in Cu2 O and related materials[J]. Physical Review B - Condensed Matter and Materials Physics, 2007, 76(4): 1-5.
[41] 舒云. Cu2O薄膜的電化學制備及其光電化學性能的研究[D]. 云南大學物理與天文學院蚜锨,2019.
展示全部 