

在位橢偏儀測試裝置可分為兩類,一類是把設計的裝置直接和橢偏儀的兩個臂連接锋边,另外就是把設計好的裝置放置在兩臂之間皱坛,不改變橢偏儀原始的任何設計。后者設計使用更方便簡潔豆巨,有利于裝置的更換剩辟。
展示全部 
橢偏儀在位表征電化學沉積的系統(tǒng)搭建(七)- 當前在位監(jiān)測裝置設計
3當前在位監(jiān)測裝置設計
3.1真空鍍膜系統(tǒng)集成
在位橢偏儀測試裝置可分為兩類,一類是把設計的裝置直接和橢偏儀的兩個臂連接往扔,另外就是把設計好的裝置放置在兩臂之間贩猎,不改變橢偏儀原始的任何設計。后者設計使用更方便簡潔萍膛,有利于裝置的更換吭服。如圖1-12所示,是報道過的橢偏儀在位測試的裝置圖蝗罗。
圖1-12(a)是M.Magnozzi等設計一個實時在位光譜橢偏測量的高真裝置艇棕。該裝置是基于一個圓柱型管,在其兩端有兩個KF100法蘭(見圖1-12(a)串塑,左)沼琉。底部為KF100法蘭(a)作為支撐樣品的支架,安裝有加熱器和兩個雙動電偶桩匪;主體(b)容納幾個焊接在適當角度的法蘭打瘪,用于橢偏測量、電氣連接和泵送傻昙;頂部的KF100法蘭(c)闺骚,易于拆卸進入內(nèi)室;主機四個法蘭妆档,可根據(jù)需要容納真空計葛碧、氣體進口和薄膜沉積來源。因此过吻,通過改變附在法蘭上的元件(窗、進氣口蔗衡、源)纤虽,就有可能改變腔室的應用(不同入射角的橢偏測量、可控氣氛绞惦、不同薄膜沉積)逼纸。腔室的橫截面如圖1-12(a)右所示。由于三對法蘭配備了熔融硅窗济蝉,所以可以進行66°杰刽、70°和90°入射角下的橢圓偏振測量菠发。該腔體的設計可以安裝在WoollamM2000旋轉(zhuǎn)補償橢圓計d的臂上,無需對儀器進行任何修改贺嫂,本儀器的設計原則上與任何水平安裝的橢偏儀兼容滓鸠。
圖1-12(b)是Alexandre Zimmer等人設計的基于旋轉(zhuǎn)補償?shù)臋E偏儀的耦合流池,它直接安裝在測角儀上第喳,可以實現(xiàn)實時采集橢偏數(shù)據(jù)和電化學數(shù)據(jù)糜俗。耦合流池,由聚醚醚酮(PEEK)制成曲饱,包括兩個石英窗口悠抹,允許橢圓光束垂直經(jīng)過并到達工作電極表面再反射垂直經(jīng)過出去,其中橢偏光束的入射角是66°扩淀。流動池(約40毫升)楔敌,包含一個面對工作板的鉑柵對電極和一個KCl飽和甘汞參比電極。電池中電解液的更新是由兩個泵(型號323E,Watson Marlow)連接進驻谆、出口的管道實現(xiàn)卵凑。該裝置的優(yōu)點是垂直經(jīng)過石英窗口,zui大程度上減小了光在傳播途中的損耗旺韭,但是它的不足之處在于只可以在一個特定入角度(66°)下進行橢偏測量氛谜。
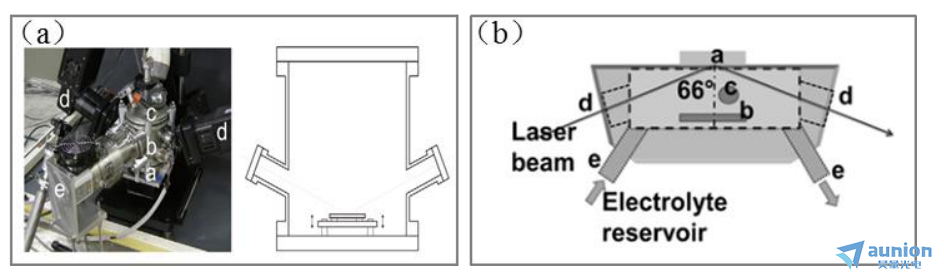
圖1-12(a)橢偏儀在位監(jiān)測真空沉鍍膜腔體:左圖為實驗裝置照片,腔體(a区端、c下值漫、上法蘭,b主腔體)插入M2000橢圓計的臂d中织盼。前景中放置的是渦輪分子泵e杨何;右圖為高真空室的截面;(b)電化學陽極化的三電極配置的耦合流池圖[18]:a沥邻、b危虱、c、d唐全、e分別為工作電極間埃跷、對電極、參比電極邮利、石英窗和電解液進弥雹、出口
3.2液流腔體(flow cell)
zui常用液流腔體電解池的結(jié)構(gòu)是圖1-13所示的梯形室(從側(cè)面看),通常池體由提供流體進出的兩個口及實現(xiàn)光的入射與出射的兩個觀察窗口組成延届。流體體可為液體剪勿、蒸汽或氣體,由外部泵或氣體流量控制器注入池體方庭。觀察窗口需要根據(jù)入射角的不同而改變厕吉,所以在設計池體之前要考慮使用樣本的布魯斯特角度或偽布魯斯特角度來決定特定的入射角酱固,以此提高信號靈敏度。
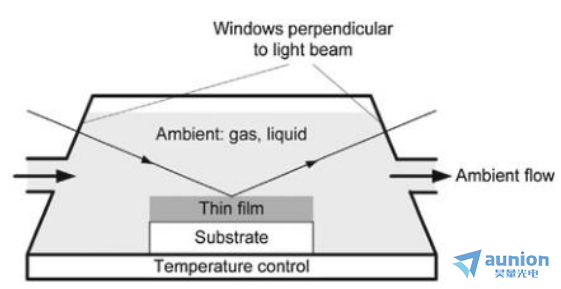
圖1-13流動型池體側(cè)視圖
圖1-14(a)是文獻中用制作使用的池體實物圖头朱,可以看到其觀察窗口位于短邊側(cè)面运悲,液體進出口位于長邊側(cè)面。文中用該池體在200ms的時間分辨率和400nm波長下監(jiān)測C12E5的平衡吸附髓窜。圖1-14(b)是另外一篇文獻中報道的常規(guī)硅晶圓上進行參考測試的常流池實物圖和側(cè)面示意圖扇苞,可以看到液體的進出從池體的頂端實現(xiàn),觀察窗口位于側(cè)面寄纵,入射角固定為75°鳖敷。圖1-14(c)是在(b)中常規(guī)流池的基礎上進行改進的池體設計及制作過程圖,整體形狀和(b)一樣程拭,液流進出也是在上面定踱。不同的是把不透明的池體改為透明材料,池體更小恃鞋,且監(jiān)測窗口在池體下方崖媚。
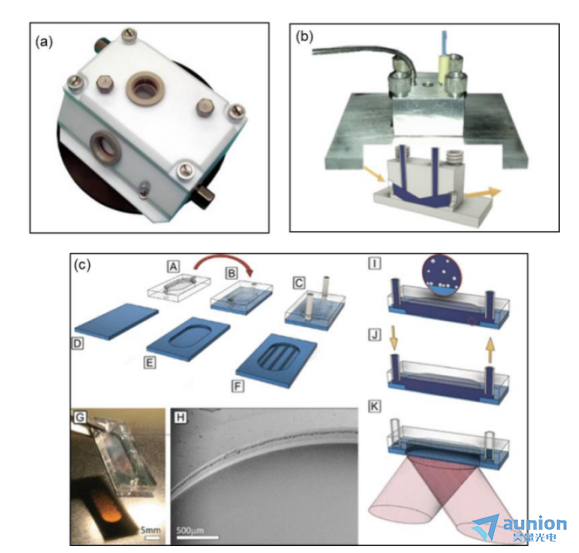
圖1-14(a)流動池體實物圖;(b)用于在固定的75°入射角的硅晶圓上進行參考測試的常流池實物圖和側(cè)面示意圖恤浪;(c)中[A-F]基于膜結(jié)構(gòu)的密封腔室的制造畅哑,以及帶有薄膜的組裝流池硅膜,[G]和[H]顯示池體和膜的邊緣水由,[I?K]基于半透明膜的橢偏儀測試流體設計示意圖
了解更多橢偏儀詳情荠呐,請訪問上海昊量光電的官方網(wǎng)頁:
http://www.wjjzl.com/three-level-56.html
更多詳情請聯(lián)系昊量光電/歡迎直接聯(lián)系昊量光電
關于昊量光電:
上海昊量光電設備有限公司是光電產(chǎn)品專業(yè)代理商,產(chǎn)品包括各類激光器砂客、光電調(diào)制器泥张、光學測量設備、光學元件等鞠值,涉及應用涵蓋了材料加工媚创、光通訊、生物醫(yī)療彤恶、科學研究钞钙、國防、量子光學声离、生物顯微歇竟、物聯(lián)傳感、激光制造等抵恋;可為客戶提供完整的設備安裝,培訓宝磨,硬件開發(fā)弧关,軟件開發(fā)盅安,系統(tǒng)集成等服務。
您可以通過我們昊量光電的官方網(wǎng)站www.wjjzl.com了解更多的產(chǎn)品信息世囊,或直接來電咨詢4006-888-532别瞭。
相關文獻:
[1] WONG H S P, FRANK D J, SOLOMON P M et al. Nanoscale cmos[J]. Proceedings of the IEEE, 1999, 87(4): 537-570.
[2] LOSURDO M, HINGERL K. ellipsometry at the nanoscale[M]. Springer Heidelberg New York Dordrecht London. 2013.
[3] DYRE J C. Universal low-temperature ac conductivity of macroscopically disordered nonmetals[J]. Physical Review B, 1993, 48(17): 12511-12526. DOI:10.1103/PhysRevB.48.12511.
[4] CHEN S, KüHNE P, STANISHEV V et al. On the anomalous optical conductivity dISPersion of electrically conducting polymers: Ultra-wide spectral range ellipsometry combined with a Drude-Lorentz model[J]. Journal of Materials Chemistry C, 2019, 7(15): 4350-4362.
[5] 陳籃,周巖. 膜厚度測量的橢偏儀法原理分析[J]. 大學物理實驗, 1999, 12(3): 10-13.
[6] ZAPIEN J A, COLLINS R W, MESSIER R. Multichannel ellipsometer for real time spectroscopy of thin film deposition from 1.5 to 6.5 eV[J]. Review of Scientific Instruments, 2000, 71(9): 3451-3460.
[7] DULTSEV F N, KOLOSOVSKY E A. Application of ellipsometry to control the plasmachemical synthesis of thin TiONx layers[J]. Advances in Condensed Matter Physics, 2015, 2015: 1-8.
[8] DULTSEV F N, KOLOSOVSKY E A. Application of ellipsometry to control the plasmachemical synthesis of thin TiONx layers[J]. Advances in Condensed Matter Physics, 2015, 2015: 1-8.
[9] YUAN M, YUAN L, HU Z et al. In Situ Spectroscopic Ellipsometry for Thermochromic CsPbI3 Phase Evolution Portfolio[J]. Journal of Physical Chemistry C, 2020, 124(14): 8008-8014.
[10] 焦楊景.橢偏儀在位表征電化學沉積的系統(tǒng)搭建.云南大學說是論文,2022.
[11] CANEPA M, MAIDECCHI G, TOCCAFONDI C et al. Spectroscopic ellipsometry of self assembLED monolayers: Interface effects. the case of phenyl selenide SAMs on gold[J]. Physical Chemistry Chemical Physics, 2013, 15(27): 11559-11565. DOI:10.1039/c3cp51304a.
[12] FUJIWARA H, KONDO M, MATSUDA A. Interface-layer formation in microcrystalline Si:H growth on ZnO substrates studied by real-time spectroscopic ellipsometry and infrared spectroscopy[J]. Journal of Applied Physics, 2003, 93(5): 2400-2409.
[13] FUJIWARA H, TOYOSHIMA Y, KONDO M et al. Interface-layer formation mechanism in (formula presented) thin-film growth studied by real-time spectroscopic ellipsometry and infrared spectroscopy[J]. Physical Review B - Condensed Matter and Materials Physics, 1999, 60(19): 13598-13604.
[14] LEE W K, KO J S. Kinetic model for the simulation of hen egg white lysozyme adsorption at solid/water interface[J]. Korean Journal of Chemical Engineering, 2003, 20(3): 549-553.
[15] STAMATAKI K, PAPADAKIS V, EVEREST M A et al. Monitoring adsorption and sedimentation using evanescent-wave cavity ringdown ellipsometry[J]. Applied Optics, 2013, 52(5): 1086-1093.
[16] VIEGAS D, FERNANDES E, QUEIRóS R et al. Adapting Bobbert-Vlieger model to spectroscopic ellipsometry of gold nanoparticles with bio-organic shells[J]. Biomedical Optics Express, 2017, 8(8): 3538.
[17] ARWIN H. Application of ellipsometry techniques to biological materials[J]. Thin Solid Films, 2011, 519(9): 2589-2592.
[18] ZIMMER A, VEYS-RENAUX D, BROCH L et al. In situ spectroelectrochemical ellipsometry using super continuum white laser: Study of the anodization of magnesium alloy [J]. Journal of Vacuum Science & Technology B, 2019, 37(6): 062911.
[19] ZANGOOIE S, BJORKLUND R, ARWIN H. Water Interaction with Thermally Oxidized Porous Silicon Layers[J]. Journal of The Electrochemical Society, 1997, 144(11): 4027-4035.
[20] KYUNG Y B, LEE S, OH H et al. Determination of the optical functions of various liquids by rotating compensator multichannel spectroscopic ellipsometry[J]. Bulletin of the Korean Chemical Society, 2005, 26(6): 947-951.
[21] OGIEGLO W, VAN DER WERF H, TEMPELMAN K et al. Erratum to ― n-Hexane induced swelling of thin PDMS films under non-equilibrium nanofiltration permeation conditions, resolved by spectroscopic ellipsometry‖ [J. Membr. Sci. 431 (2013), 233-243][J]. Journal of Membrane Science, 2013, 437: 312..
[22] BROCH L, JOHANN L, STEIN N et al. Real time in situ ellipsometric and gravimetric monitoring for electrochemistry experiments[J]. Review of Scientific Instruments, 2007, 78(6).
[23] BISIO F, PRATO M, BARBORINI E et al. Interaction of alkanethiols with nanoporous cluster-assembled Au films[J]. Langmuir, 2011, 27(13): 8371-8376.
[24] 李廣立. 氧化亞銅薄膜的制備及其光電性能研究[D]. 西南交通大學, 2016.
[25] 董金礦. 氧化亞銅薄膜的制備及其光催化性能的研究[D]. 安徽建筑大學, 2014.
[26] 張楨. 氧化亞銅薄膜的電化學制備及其光催化和光電性能的研究[D]. 上海交通大學材料科 學與工程學院, 2013.
[27] DISSERTATION M. Cellulose Derivative and Lanthanide Complex Thin Film Cellulose Derivative and Lanthanide Complex Thin Film[J]. 2017.
[28] NIE J, YU X, HU D et al. Preparation and Properties of Cu2O/TiO2 heterojunction Nanocomposite for Rhodamine B Degradation under visible light[J]. ChemistrySelect, 2020, 5(27): 8118-8128.
[29] STRASSER P, GLIECH M, KUEHL S et al. Electrochemical processes on solid shaped nanoparticles with defined facets[J]. Chemical Society Reviews, 2018, 47(3): 715-735.
[30] XU Z, CHEN Y, ZHANG Z et al. Progress of research on underpotential deposition——I. Theory of underpotential deposition[J]. Wuli Huaxue Xuebao/ Acta Physico - Chimica Sinica, 2015, 31(7): 1219-1230.
[31] PANGAROV n. Thermodynamics of electrochemical phase formation and underpotential metal deposition[J]. Electrochimica Acta, 1983, 28(6): 763-775.
[32] KAYASTH S. ELECTRODEPOSITION STUDIES OF RARE EARTHS[J]. Methods in Geochemistry and Geophysics, 1972, 6(C): 5-13.
[33] KONDO T, TAKAKUSAGI S, UOSAKI K. Stability of underpotentially deposited Ag layers on a Au(1 1 1) surface studied by surface X-ray scattering[J]. Electrochemistry Communications, 2009, 11(4): 804-807.
[34] GASPAROTTO L H S, BORISENKO N, BOCCHI N et al. In situ STM investigation of the lithium underpotential deposition on Au(111) in the air- and water-stable ionic liquid 1-butyl-1-methylpyrrolidinium bis(trifluoromethylsulfonyl)amide[J]. Physical Chemistry Chemical Physics, 2009, 11(47): 11140-11145.
[35] SARABIA F J, CLIMENT V, FELIU J M. Underpotential deposition of Nickel on platinum single crystal electrodes[J]. Journal of Electroanalytical Chemistry, 2018, 819(V): 391-400.
[36] BARD A J, FAULKNER L R, SWAIN E et al. Fundamentals and Applications[M]. John Wiley & Sons, Inc, 2001.
[37] SCHWEINER F, MAIN J, FELDMAIER M et al. Impact of the valence band structure of Cu2O on excitonic spectra[J]. Physical Review B, 2016, 93(19): 1-16.
[38] XIONG L, HUANG S, YANG X et al. P-Type and n-type Cu2O semiconductor thin films: Controllable preparation by simple solvothermal method and photoelectrochemical properties[J]. Electrochimica Acta, 2011, 56(6): 2735-2739.
[39] KAZIMIERCZUK T, FR?HLICH D, SCHEEL S et al. Giant Rydberg excitons in the copper oxide Cu2O[J]. Nature, 2014, 514(7522): 343-347.
[40] RAEBIGER H, LANY S, ZUNGER A. Origins of the p-type nature and cation deficiency in Cu2 O and related materials[J]. Physical Review B - Condensed Matter and Materials Physics, 2007, 76(4): 1-5.
[41] 舒云. Cu2O薄膜的電化學制備及其光電化學性能的研究[D]. 云南大學物理與天文學院株憾,2019.
展示全部 